 |
The
Flagellum Unspun
The Collapse of "Irreducible Complexity"
Kenneth
R. Miller
Brown University
Providence, Rhode Island 02912 USA
This
is a pre-publication copy of an article that appeared in "Debating Design from Darwin to DNA," edited by Michael Ruse and William Dembski. Debating Design is available from Amazon.com.
|
Almost from the moment The Origin of Species was published in 1859, the opponents of evolution have fought a long, losing
battle against their Darwinian foes. Today, like a prizefighter in the late
rounds losing badly on points, they've placed their hopes in one big punch
– a single claim that might smash through the overwhelming weight of
scientific evidence to bring Darwin to the canvas once and for all. Their
name for this virtual roundhouse right is "intelligent design."
In the last several years, the intelligent design movement
has attempted to move against science education standards in several American
states, most famously in Kansas and Ohio (Holden 1999; Gura 2002). The principal
claim made by adherents of this view is that they can detect the presence
of "intelligent design" in complex biological systems. As evidence, they cite
a number of specific examples, including the vertebrate blood clotting cascade,
the eukaryotic cilium, and most notably, the eubacterial flagellum (Behe 1996a,
Behe 2002).
Of all these examples, the flagellum has been presented
so often as a counter-example to evolution that it might well be considered
the "poster child" of the modern anti-evolution movement. Variations of its
image (Figure 1) now appear on web pages of anti-evolution groups like the
Discovery Institute, and on the covers of "intelligent design" books such
as William Dembski's No Free Lunch (Dembski 2002a). To anti-evolutionists,
the high status of the flagellum reflects the supposed fact that it could
not possibly have been produced by an evolutionary pathway.
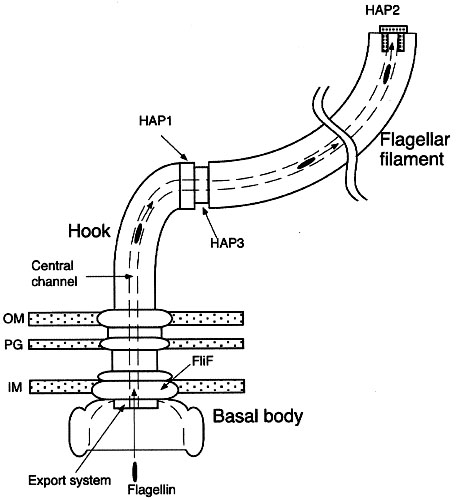
Figure 1:
The eubacterial flagellum. The flagellum is an ion-powered rotary motor,
anchored in the membranes surrounding the bacterial cell. This schematic
diagram highlights the assembly process of the bacterial flagellar filament
and the cap-filament complex. OM, outer membrane; PG, peptidoglycan layer;
IM, cytoplasmic membrane (From Yonekura et al 2000).
There is, to be sure, nothing new or novel in an anti-evolutionist
pointing to a complex or intricate natural structure, and professing skepticism
that it could have been produced by the "random" processes of mutation and
natural selection. Nonetheless, the "argument from personal incredulity,"
as such sentiment has been appropriately described, has been a weapon of little
value in the anti-evolution movement. Anyone can state at any time that they
cannot imagine how evolutionary mechanisms might have produced a certain species,
organ, structure. Such statements, obviously, are personal – and they
say more about the limitations of those who make them than they do about the
limitations of Darwinian mechanisms.
The hallmark of the intelligent design movement, however,
is that it purports to rise above the level of personal skepticism. It claims
to have found a reason why evolution could not have produced a structure
like the bacterial flagellum, a reason based on sound, solid scientific evidence.
Why does the intelligent design movement regard the
flagellum as unevolvable? Because it is said to possesses a quality known
as "irreducible complexity." Irreducibly complex structures, we are told,
could not have been produced by evolution, or, for that matter, by any natural
process. They do exist, however, and therefore they must have been produced
by something. That something could only be an outside intelligent agency operating
beyond the laws of nature – an intelligent designer. That, simply stated,
is the core of the new argument from design, and the intellectual basis of
the intelligent design movement.
The great irony of the flagellum's increasing acceptance
as an icon of anti-evolution is that fact that research had demolished its
status as an example of irreducible complexity almost at the very moment it
was first proclaimed. The purpose of this article is to explore the arguments
by which the flagellum's notoriety has been achieved, and to review the research
developments that have now undermined they very foundations of those arguments.
The Argument's Origins
The flagellum owes its status principally to Darwin's
Black Box (Behe 1996a) a book by Michael Behe that employed it in a carefully-crafted
anti-evolution argument. Building upon William Paley's well-known "argument
from design," Behe sought to bring the argument two centuries forward into
the realm of biochemistry. Like Paley, Behe appealed to his readers to appreciate
the intricate complexity of living organisms as evidence for the work of a
designer. Unlike Paley, however, he raised the argument to a new level, claiming
to have discovered a scientific principle that could be used to prove that
certain structures could not have been produced by evolution. That principle
goes by the name of "irreducible complexity."
An irreducibly complex structure is defined as ". .
. a single system composed of several well-matched, interacting parts that
contribute to the basic function, wherein the removal of any one of the parts
causes the system to effectively cease functioning." (Behe 1996a, 39) Why
would such systems present difficulties for Darwinism? Because they could
not possibly have been produced by the process of evolution:
"An irreducibly complex system cannot be produced
directly by numerous, successive, slight modifications of a precursor
system, because any precursor to an irreducibly complex system that is
missing a part is by definition nonfunctional. .... Since natural selection
can only choose systems that are already working, then if a biological
system cannot be produced gradually it would have to arise as an integrated
unit, in one fell swoop, for natural selection to have anything to act
on." (Behe 1996b)
The phrase "numerous, successive, slight modifications"
is not accidental. The very same words were used by Charles Darwin in The
Origin of Species in describing the conditions that had to be met for
his theory to be true. As Darwin wrote, if one could find an organ or structure
that could not have been formed by "numerous, successive, slight modifications,"
his "theory would absolutely break down" (Darwin 1859, 191). To anti-evolutionists,
the bacterial flagellum is now regarded as exactly such a case – an "irreducibly
complex system" which "cannot be produced directly by numerous successive,
slight modifications." A system that could not have evolved – a desperation
punch that just might win the fight in the final round – a tool with
which the theory of evolution can be brought down.
The Logic of Irreducible Complexity
Living cells are filled, of course, with complex structures
whose detailed evolutionary origins are not known. Therefore, in fashioning
an argument against evolution one might pick nearly any cellular structure,
the ribosome for example, and claim – correctly – that its origin
has not been explained in detail by evolution.
Such arguments are easy to make, of course, but nature
of scientific progress renders them far from compelling. The lack of a detailed
current explanation for a structure, organ, or process does not mean that
science will never come up with one. As an example, one might consider the
question of how left-right asymmetry arises in vertebrate development, a question
that was beyond explanation until the 1990s (Belmonte 1999). In 1990 one might
have argued that the body's left-right asymmetry could just as well be explained
by the intervention of a designer as by an unknown molecular mechanism. Only
a decade later, the actual molecular mechanism was identified (Stern 2002),
and any claim one might have made for the intervention of a designer would
have been discarded. The same point can be made, of course, regarding any
structure or mechanism whose origins are not yet understood.
The utility of the bacterial flagellum is that it seems
to rise above this "argument from ignorance." By asserting that it is a structure
"in which the removal of an element would cause the whole system to cease
functioning" (Behe 2002), the flagellum is presented as a "molecular machine"
whose individual parts must have been specifically crafted to work as a unified
assembly. The existence of such a multipart machine therefore provides genuine
scientific proof of the actions of an intelligent designer.
In the case of the flagellum, the assertion of irreducible
complexity means that a minimum number of protein components, perhaps 30,
are required to produce a working biological function. By the logic of irreducible
complexity, these individual components should have no function until all
30 are put into place, at which point the function of motility appears. What
this means, of course, is that evolution could not have fashioned those components
a few at a time, since they do not have functions that could be favored by
natural selection. As Behe wrote: " . . . natural selection can only choose
among systems that are already working" (Behe 2002), and an irreducibly complex
system does not work unless all of its parts are in place. The flagellum is
irreducibly complex, and therefore, it must have been designed. Case closed.
Answering the Argument
The assertion that cellular machines are irreducibly
complex, and therefore provide proof of design, has not gone unnoticed by
the scientific community. A number of detailed rebuttals have appeared in
the literature, and many have pointed out the poor reasoning of recasting
the classic argument from design in the modern language of biochemistry (Coyne
1996; Miller 1996; Depew 1998; Thornhill and Ussery 2000). I have suggested
elsewhere that the scientific literature contains counter-examples to any
assertion that evolution cannot explain biochemical complexity (Miller 1999,
147), and other workers have addressed the issue of how evolutionary mechanisms
allow biological systems to increase in information content (Schneider 2000;
Adami, Ofria, and Collier 2000).
The most powerful rebuttals to the flagellum story,
however, have not come from direct attempts to answer the critics of evolution.
Rather, they have emerged from the steady progress of scientific work on the
genes and proteins associated with the flagellum and other cellular structures.
Such studies have now established that the entire premise by which this molecular
machine has been advanced as an argument against evolution is wrong –
the bacterial flagellum is not irreducibly complex. As we will see,
the flagellum – the supreme example of the power of this new "science
of design" – has failed its most basic scientific test. Remember the
claim that "any precursor to an irreducibly complex system that is missing
a part is by definition nonfunctional?" As the evidence has shown, nature
is filled with examples of "precursors" to the flagellum that are indeed "missing
a part," and yet are fully-functional. Functional enough, in some cases, to
pose a serious threat to human life.
The Type -III Secretory Apparatus
In the popular imagination, bacteria are "germs" –
tiny microscopic bugs that make us sick. Microbiologists smile at that generalization,
knowing that most bacteria are perfectly benign, and many are beneficial –
even essential – to human life. Nonetheless, there are indeed bacteria
that produce diseases, ranging from the mildly unpleasant to the truly dangerous.
Pathogenic, or disease-causing, bacteria threaten the organisms they infect
in a variety of ways, one of which is to produce poisons and inject them directly
into the cells of the body. Once inside, these toxins break down and destroy
the host cells, producing illness, tissue damage, and sometimes even death.
In order to carry out this diabolical work, bacteria
must not only produce the protein toxins that bring about the demise of their
hosts, but they must efficiently inject them across the cell membranes and
into the cells of their hosts. They do this by means of any number of specialized
protein secretory systems. One, known as the type III secretory system (TTSS),
allows gram negative bacteria to translocate proteins directly into the cytoplasm
of a host cell (Heuck 1998). The proteins transferred through the TTSS include
a variety of truly dangerous molecules, some of which are known as "virulence
factors," and are directly responsible for the pathogenic activity of some
of the most deadly bacteria in existence (Büttner and Bonas 2002; Heuck
1998).
At first glance, the existence of the TTSS, a nasty
little device that allows bacteria to inject these toxins through the cell
membranes of its unsuspecting hosts, would seem to have little to do with
the flagellum. However, molecular studies of proteins in the TTSS have revealed
a surprising fact – the proteins of the TTSS are directly homologous
to the proteins in the basal portion of the bacterial flagellum. As figure
2 (Heuck 1998) shows, these homologies extend to a cluster of closely-associated
proteins found in both of these molecular "machines." On the basis of these
homologies, McNab (McNab 1999) has argued that the flagellum itself should
be regarded as a type III secretory system. Extending such studies with a
detailed comparison of the proteins associated with both systems, Aizawa has
seconded this suggestion, noting that the two systems "consist of homologous
component proteins with common physico-chemical properties" (Aizawa 2001,
163). It is now clear, therefore, that a smaller subset of the full complement
of proteins in the flagellum makes up the functional transmembrane portion
of the TTSS.

Figure 2:
There are extensive homologies between type III secretory proteins and
proteins involved in export in the basal region of the bacterial flagellum.
These homologies demonstrate that the bacterial flagellum is not "irreducibly
complex." In this diagram (redrawn from Heuck 1998), the shaded portions
of the basal region indicate proteins in the E. coli flagellum
homologous to the Type III secretory structure of Yersinia. . OM,
outer membrane; PP, periplasmic space; CM, cytoplasmic membrane.
Stated directly, the TTSS does its dirty work using
a handful of proteins from the base of the flagellum. From the evolutionary
point of view, this relationship is hardly surprising. In fact, it's to be
expected that the opportunism of evolutionary processes would mix and match
proteins to produce new and novel functions. According to the doctrine of
irreducible complexity, however, this should not be possible. If the flagellum
is indeed irreducibly complex, then removing just one part, let alone 10 or
15, should render what remains "by definition nonfunctional." Yet the TTSS
is indeed fully-functional, even though it is missing most of the parts of
the flagellum. The TTSS may be bad news for us, but for the bacteria that
possess it, it is a truly valuable biochemical machine.
The existence of the TTSS in a wide variety of bacteria
demonstrates that a small portion of the "irreducibly complex" flagellum can
indeed carry out an important biological function. Since such a function is
clearly favored by natural selection, the contention that the flagellum must
be fully-assembled before any of its component parts can be useful is obviously
incorrect. What this means is that the argument for intelligent design of
the flagellum has failed.
Counterattack
Classically, one of the most widely-repeated charges
made by anti-evolutionists is that the fossil record contains wide "gaps"
for which transitional fossils have never been found. Therefore, the intervention
of a creative agency, an intelligent designer, must be invoked to account
for each gap. Such gaps, of course, have been filled with increasing frequency
by paleontologists – the increasingly rich fossil sequences demonstrating
the origins of whales are a useful examples (Thewissen, Hussain, and Arif
1994; Thewissen, Williams, Roe, and Hussain 2001). Ironically, the response
of anti-evolutionists to such discoveries is frequently to claim that things
have only gotten worse for evolution. Where previously there had been just
one gap, as a result of the transitional fossil, now there are two (one on
either side of the newly-discovered specimen).
As word of the relationship between the eubacterial
flagellum and the TTSS has begun to spread among the "design" community, the
first hints of a remarkably similar reaction have emerged. The TTSS only makes
problems worse for evolution, according to this response, because now there
are two irreducibly-complex systems to deal with. The flagellum is still irreducibly
complex – but so is the TTSS. But now there are two systems for evolutionists
to explain instead of just one.
Unfortunately for this line of argument, the claim
that one irreducibly-complex system might contain another is self-contradictory.
To understand this, we need to remember that the entire point of the design
argument, as exemplified by the flagellum, is that only the entire biochemical
machine, with all of its parts, is functional. For the intelligent design
argument to stand, this must be the case, since it provides the basis for
their claim that only the complete flagellum can be favored by natural selection,
not any its component parts.
However, if the flagellum contains within it a smaller
functional set of components like the TTSS, then the flagellum itself cannot
be irreducibly complex – by definition. Since we now know that this is
indeed the case, it is obviously true that the flagellum is not irreducibly
complex.
A second reaction, which I have heard directly after
describing the relationship between the secretory apparatus and the flagellum,
is the objection that the TTSS does not tell us how either it or the flagellum
evolved. This is certainly true, although Aizawa has suggested that the TTSS
may indeed be an evolutionary precursor of the flagellum (Aizawa 2001). Nonetheless,
until we have produced a step-by-step account for the evolutionary derivation
of the flagellum, one may indeed invoke the argument from ignorance for this
and every other complex biochemical machine.
However, in agreeing to this, one must keep in mind
that the doctrine of irreducible complexity was intended to go one step beyond
the claim of ignorance. It was fashioned in order to provide a rationale for
claiming that the bacterial flagellum couldn't have evolved, even in principle,
because it is irreducibly complex. Now that a simpler, functional system (the
TTSS) has been discovered among the protein components of the flagellum, the
claim of irreducible complexity has collapsed, and with it any "evidence"
that the flagellum was designed.
Combinatorial Argument
At first glance, William Dembski's case for intelligent
design seems to follow a distinctly different strategy in dealing with biological
complexity. His recent book, No Free Lunch (Dembski 2002a), lays out
this case, using information theory and mathematics to show that life is the
result of intelligent design. Dembski makes the assertion that living organisms
contain what he calls "complex specified information" (CSI), and claims to
have shown that the evolutionary mechanism of natural selection cannot produce
CSI. Therefore, any instance of CSI in a living organism must be the result
of intelligent design. And living organisms, according to Dembski, are chock-full
of CSI.
Dembski's arguments, couched in the language of information
theory, are highly technical and are defended, almost exclusively, by reference
to their utility in detecting information produced by human beings. These
include phone and credit card numbers, symphonies, and artistic woodcuts,
to name just a few. One might then expect that Dembski, having shown how the
presence of CSI can be demonstrated in man made objects, would then turn to
a variety of biological objects. Instead, he turns to just one such object,
the bacterial flagellum.
Dembski then offers his readers a calculation showing
that the flagellum could not have possibly have evolved. Significantly, he
begins that calculation by linking his arguments to those of Behe, writing:
"I want therefore in this section to show how irreducible complexity is a
special case of specified complexity, and in particular I want to sketch how
one calculates the relevant probabilities needed to eliminate chance and infer
design for such systems" (Dembski 2002a, 289). Dembski then tells us that
an irreducibly complex system, like the flagellum, is a "discrete combinatorial
object." What this means, as he explains, is that the probability of assembling
such an object can be calculated by determining the probabilities that each
of its components might have originated by chance, that they might have been
localized to the same region of the cell, and that they would be assembled
in precisely the right order. Dembski refers to these three probabilities
as Porig,
Plocal,
and Pconfig,
and he regards each of them as separate and independent (Dembski 2002a, 291).
This approach overlooks the fact that the last two
probabilities are actually contained within the first. Localization and self-assembly
of complex protein structures in prokaryotic cells are properties generally
determined by signals built into the primary structures of the proteins themselves.
The same is likely true for the amino acid sequences of the 30 or so protein
components of the flagellum and the approximately 20 proteins involved in
the flagellum's assembly (McNab 1999; Yonekura et al 2000). Therefore,
if one gets the sequences of all the proteins right, localization and assembly
will take care of themselves.
To the ID enthusiast, however, this is a point of little
concern. According to Dembski, evolution could still not construct the 30
proteins needed for the flagellum. His reason is that the probability of their
assembly falls below what he terms the "universal probability bound." According
to Dembski, the probability bound is a sensible allowance for the fact that
highly improbable events do occur from time to time in nature. To allow for
such events, he agrees that given enough time, any event with a probability
larger than 10-150
might well take place. Therefore, if a sequence of events, such as a presumed
evolutionary pathway, has a calculated probability less than 10-150
, we may conclude that the pathway is impossible. If the calculated probability
is greater than 10-150,
it's possible (even if unlikely).
When Dembski turns his attention to the chances of
evolving the 30 proteins of the bacterial flagellum, he makes what he regards
as a generous assumption. Guessing that each of the proteins of the flagellum
have about 300 amino acids, one might calculate that the chances of getting
just one such protein to assemble from "random" evolutionary processes would
be 20-300
, since there are 20 amino acids specified by the genetic code. Dembski, however,
concedes that proteins need not get the exact amino acid sequence right
in order to be functional, so he cuts the odds to just 20-30,
which he tells his readers is "on the order of 10-39"
(Dembski 2002a, 301). Since the flagellum requires
30 such proteins, he explains that 30 such probabilities "will all need to
be multiplied to form the origination probability"(Dembski 2002a, 301). That
would give us an origination probability for the flagellum of 10 -1170,
far below the universal probability bound. The flagellum couldn't have evolved,
and now we have the numbers to prove it. Right?
Assuming Impossibility
I have no doubt that to the casual reader, a quick
glance over the pages of numbers and symbols in Dembski's books is impressive,
if not downright intimidating. Nonetheless, the way in which he calculates
the probability of an evolutionary origin for the flagellum shows how little
biology actually stands behind those numbers. His computation calculates only
the probability of spontaneous, random assembly for each of the proteins of
the flagellum. Having come up with a probability value on the order of 10
-1170, he
assures us that he has shown the flagellum to be unevolvable. This conclusion,
of course, fits comfortably with his view is that "The Darwinian mechanism
is powerless to produce irreducibly complex systems..." (Dembski 2002a, 289).
However complex Dembski's analysis, the scientific
problem with his calculations is almost too easy to spot. By treating the
flagellum as a "discrete combinatorial object" he has shown only that it is
unlikely that the parts flagellum could assemble spontaneously. Unfortunately
for his argument, no scientist has ever proposed that the flagellum or any
other complex object evolved that way. Dembski, therefore, has constructed
a classic "straw man" and blown it away with an irrelevant calculation.
By treating the flagellum as a discrete combinatorial
object he has assumed in his calculation that no subset of the 30 or so proteins
of the flagellum could have biological activity. As we have already seen,
this is wrong. Nearly a third of those proteins are closely related to components
of the TTSS, which does indeed have biological activity. A calculation that
ignores that fact has no scientific validity.
More importantly, Dembski's willingness to ignore the
TTSS lays bare the underlying assumption of his entire approach towards the
calculation of probabilities and the detection of "design." He assumes
what he is trying to prove.
According to Dembski, the detection of "design" requires
that an object display complexity that could not be produced by what he calls
"natural causes." In order to do that, one must first examine all of the possibilities
by which an object, like the flagellum, might have been generated naturally.
Dembski and Behe, of course, come to the conclusion that there are no such
natural causes. But how did they determine that? What is the scientific method
used to support such a conclusion? Could it be that their assertions of the
lack of natural causes simply amount to an unsupported personal belief? Suppose
that there are such causes, but they simply happened not to think of them?
Dembski actually seems to realize that this is a serious problem. He writes:
"Now it can happen that we may not know enough to determine all the relevant
chance hypotheses [which here, as noted above, means all relevant natural
processes (hvt)]. Alternatively, we might think we know the relevant chance
hypotheses, but later discover that we missed a crucial one. In the one case
a design inference could not even get going; in the other, it would be mistaken"
(Dembski 2002, 123 (note 80)).
What Dembski is telling us is that in order to "detect"
design in a biological object one must first come to the conclusion that the
object could not have been produced by any "relevant chance hypotheses" (meaning,
naturally, evolution). Then, and only then, are Dembski's calculations brought
into play. Stated more bluntly, what this really means is that the "method"
first involves assuming the absence of an evolutionary pathway leading
to the object, followed by a calculation "proving" the impossibility of spontaneous
assembly. Incredibly, this a priori reasoning is exactly the sort of
logic upon which the new "science of design" has been constructed.
Not surprisingly, scientific reviewers have not missed
this point – Dembski's arguments have been repeatedly criticized on this
issue and on many others (Orr 2002; Charlesworth 2002; Padian 2002).
Designing the Cycle
In assessing the design argument, therefore, it only
seems as though two distinct arguments have been raised for the unevolvability
of the flagellum. In reality, those two arguments, one invoking irreducible
complexity and the other specified complex information, both depend upon a
single scientifically insupportable position. Namely, that we can look at
a complex biological object and determine with absolute certainty that none
of its component parts could have been first selected to perform other functions.
The discovery of extensive homologies between the Type III secretory system
and the flagellum has now shown just how wrong that position was.
When anti-evolutionary arguments featuring the bacterial
flagellum rose into prominence, beginning with the 1996 publication of Darwin's
Black Box (Behe 1996a), they were predicated upon the assertion that each
of the protein components of the flagellum were crafted, in a single act of
design, to fit the specific purpose of the flagellum. The flagellum was said
to be unevolvable since the entire complex system had to be assembled first
in order to produce any selectable biological function. This claim was broadened
to include all complex biological systems, and asserted further that science
would never find an evolutionary pathway to any of these systems. After all,
it hadn't so far, at least according to one of "design's" principal advocates:
There is no publication in the scientific
literature – in prestigious journals, specialty journals, or books
– that describes how molecular evolution of any real, complex, biochemical
system either did occur or even might have occurred.
(Behe 1996a, 185)
As many critics of intelligent design have pointed
out, that statement is simply false. Consider, as just one example, the Krebs
cycle, an intricate biochemical pathway consisting of nine enzymes and a number
of cofactors that occupies center stage in the pathways of cellular metabolism.
The Krebs cycle is "real," "complex," and "biochemical." Does it also present
a problem for evolution? Apparently yes, according to the authors of a 1996
paper in the Journal of Molecular evolution, who wrote:
"The Krebs cycle has been frequently quoted
as a key problem in the evolution of living cells, hard to explain by
Darwin’s natural selection: How could natural selection explain the
building of a complicated structure in toto, when the intermediate stages
have no obvious fitness functionality? (Melendez-Hevia,
Wadell, and Cascante 1996)
Where intelligent design theorists throw up their hands
and declare defeat for evolution, however, these researchers decided to do
the hard scientific work of analyzing the components of the cycle, and seeing
if any of them might have been selected for other biochemical tasks. What
they found should be a lesson to anyone who asserts that evolution can only
act by direct selection for a final function. In fact, nearly all of the proteins
of the complex cycle can serve different biochemical purposes within the cell,
making it possible to explain in detail how they evolved:
In the Krebs cycle problem the intermediary
stages were also useful, but for different purposes, and, therefore, its
complete design was a very clear case of opportunism. . . . the Krebs
cycle was built through the process that Jacob (1977) called ‘‘evolution
by molecular tinkering,’’ stating that evolution does not produce
novelties from scratch: It works on what already exists. The most novel
result of our analysis is seeing how, with minimal new material, evolution
created the most important pathway of metabolism, achieving the best chemically
possible design. In this case, a chemical engineer who was looking for
the best design of the process could not have found a better design than
the cycle which works in living cells." (Melendez-Hevia,
Wadell, and Cascante 1996)
Since this paper appeared, a study based on genomic
DNA sequences has confirmed the validity of this approach (Huynen, Dandekar,
and Bork 1999). By contrast, how would intelligent design have approached
the Krebs Cycle? Using Dembski's calculations as our guide, we would first
determine the amino acid sequences of each of the proteins of the cycle, and
then calculate the probability of their spontaneous assembly. When this is
done, an origination probability of less than 10 -400
is the result. Therefore, the result of applying "design" as a predictive
science would have told both groups of researchers that their ultimately successful
studies would have been fruitless, since the probability of spontaneous assembly
falls below the "universal probability bound."
We already know, however, the reason that such calculations
fail. They carry a built-in assumption that the component parts of a complex
biochemical system have no possible functions beyond the completely assembled
system itself. As we have seen, this assumption is false. The Krebs cycle
researchers knew better, of course, and were able to produce two important
studies describing how a real, complex, biochemical system might have evolved
– the very thing that design theorists once claimed did not exist in
the scientific literature.
The Failure of Design
It is no secret that concepts like "irreducible complexity"
and "intelligent design" have failed to take the scientific community by storm
(Forrest 2002). Design has not prompted new research studies, new breakthroughs,
or novel insights on so much as a single scientific question. Design advocates
acknowledge this from time to time, but they often claim that this is because
the scientific deck is stacked against them. The Darwinist establishment,
they say, prevents them from getting a foot in the laboratory door.
I would suggest that the real reason for the cold shoulder
given "design" by the scientific community, particularly by life science researchers,
is because time and time again its principal scientific claims have turned
out to be wrong. Science is a pragmatic activity, and if your hypothesis doesn't
work, it is quickly discarded.
The claim of irreducible complexity for the bacterial
flagellum is an obvious example of this, but there are many others. Consider,
for example, the intricate cascade of proteins involved in the clotting of
vertebrate blood. This has been cited as one of the principal examples of
the kind of complexity that evolution cannot generate, despite the elegant
work of Russell Doolittle (Doolittle and Feng 1987; Doolittle 1993) to the
contrary. A number of proteins are involved in this complex pathway, as described
by Behe:
When an animal is cut, a protein called Hagemann
factor (XII) sticks to the surface of cells near the wound. Bound Hagemann
factor is then cleaved by a protein called HMK to yield activated Hagemann
factor. Immediately the activated Hagemann factor converts another protein,
called prekallikrein, to its active form, kallikrein.
(Behe 1996a, 84)
How important are each of these proteins? In line with
the dogma of irreducible complexity, Behe argues that each and every component
must be in place before the system will work, and he is perfectly clear on
this point:
. . . none of the cascade proteins are used
for anything except controlling the formation of a clot. Yet in the absence
of any of the components, blood does not clot, and the system fails. (Behe
1996a, 86)
As we have seen, the claim that every one of the components
must be present for clotting to work is central to the "evidence" for design.
One of those components, as these quotations indicate, is Factor XII, which
initiates the cascade. Once again, however, a nasty little fact gets in the
way of intelligent design theory. Dolphins lack Factor XII (Robinson, Kasting,
and Aggeler 1969), and yet their blood clots perfectly well. How can this
be if the clotting cascade is indeed irreducibly complex? It cannot, of course,
and therefore the claim of irreducible complexity is wrong for this system
as well. I would suggest, therefore, that the real reason for the rejection
of "design" by the scientific community is remarkably simple – the claims
of the intelligent design movement are contradicted time and time again by
the scientific evidence.
The Flagellum Unspun
In any discussion of the question of "intelligent design,"
it is absolutely essential to determine what is meant by the term itself.
If, for example, the advocates of design wish to suggest that the intricacies
of nature, life, and the universe reveal a world of meaning and purpose consistent
with an overarching, possibly Divine intelligence, then their point is philosophical,
not scientific. It is a philosophical point of view, incidentally, that I
share, along with many scientists. As H. Allen Orr pointed out in a recent
review:
Plenty of scientists have, after all, been
attracted to the notion that natural laws reflect (in some way that's
necessarily poorly articulated) an intelligence or aesthetic sensibility.
This is the religion of Einstein, who spoke of "the grandeur of reason
incarnate in existence" and of the scientist's "religious feeling [that]
takes the form of a rapturous amazement at the harmony of natural law."
(Orr 2002).
This, however, is not what is meant by "intelligent
design" in the parlance of the new anti-evolutionists. Their views demand
not a universe in which the beauty and harmony of natural law has brought
a world of vibrant and fruitful life into existence, but rather a universe
in which the emergence and evolution of life is made expressly impossible
by the very same rules. Their view requires that the source of each and every
novelty of life was the direct and active involvement of an outside designer
whose work violated the very laws of nature he had fashioned. The world of
intelligent design is not the bright and innovative world of life that we
have come to know through science. Rather, it is a brittle and unchanging
landscape, frozen in form and unable to adapt except at the whims of its designer.
Certainly, the issue of design and purpose in nature
is a philosophical one that scientists can and should discuss with great vigor.
However, the notion at the heart's of today intelligent design movement is
that the direct intervention of an outside designer can be demonstrated by
the very existence of complex biochemical systems. What even they acknowledge
is that their entire scientific position rests upon a single assertion –
that the living cell contains biochemical machines that are irreducibly complex.
And the bacterial flagellum is the prime example of such a machine.
Such an assertion, as we have seen, can be put to the
test in a very direct way. If we are able to search and find an example of
a machine with fewer protein parts, contained within the flagellum, that serves
a purpose distinct from motility, the claim of irreducible complexity is refuted.
As we have also seen, the flagellum does indeed contain such a machine, a
protein-secreting apparatus that carries out an important function even in
species that lack the flagellum altogether. A scientific idea rises or falls
on the weight of the evidence, and the evidence in the case of the bacterial
flagellum is abundantly clear.
As an icon of anti-evolution, the flagellum has fallen.
The very existence of the Type III Secretory System
shows that the bacterial flagellum is not irreducibly complex. It also demonstrates,
more generally, that the claim of "irreducible complexity" is scientifically
meaningless, constructed as it is upon the flimsiest of foundations –
the assertion that because science has not yet found selectable functions
for the components of a certain structure, it never will. In the final analysis,
as the claims of intelligent design fall by the wayside, its advocates are
left with a single, remaining tool with which to battle against the rising
tide of scientific evidence. That tool may be effective in some circles, of
course, but the scientific community will be quick to recognize it for what
it really is – the classic argument from ignorance, dressed up in the
shiny cloth of biochemistry and information theory.
When three leading advocates of intelligent design
were recently given a chance to make their case in an issue of Natural
History magazine, they each concluded their articles with a plea for design.
One wrote that we should recognize "the design inherent in life and the universe"
(Behe 2002), another that "design remains a possibility" (Wells 2002), and
another "that the natural sciences need to leave room for design" (Dembski
2002b). Yes, it is true. Design does remain a possibility, but not the type
of "intelligent design" of which they speak.
As Darwin wrote, there is grandeur in an evolutionary
view of life, a grandeur that is there for all to see, regardless of their
philosophical views on the meaning and purpose of life. I do not believe,
even for an instant, that Darwin's vision has weakened or diminished the sense
of wonder and awe that one should feel in confronting the magnificence and
diversity of the living world. Rather, to a person of faith it should enhance
their sense of the Creator's majesty and wisdom (Miller 1999). Against such
a backdrop, the struggles of the intelligent design movement are best understood
as clamorous and disappointing double failures – rejected by science
because they do not fit the facts, and having failed religion because they
think too little of God.
Bibliography
Adami, C., C. Ofria, and T. C. Collier, 2000. Evolution
of biological complexity, Proceedings of the National Academy of Sciences
97: 4463—4468.
Aizawa, S.-I., 2001. Bacterial flagella and
type III secretion systems, FEMS Microbiology Letters 202: 157-164.
Behe, M, 1996b. Evidence for Intelligent Design
from Biochemistry, a speech given at the Discovery Institute's God &
Culture Conference, August 10, 1996 Seattle, WA. Available from World
Wide Web: http://www.arn.org/docs/behe/mb_idfrombiochemistry.htm
Behe, M. 1996a. Darwin's Black Box. New
York: The Free Press.
Behe, M., 2002. The challenge of irreducible complexity,
Natural History 111 (April): 74.
Büttner D., and U. Bonas, 2002. Port of
entry - the Type III secretion translocon, Trends in Microbiology
10: 186-191.
Belmonte, J. C. I. , 1999. How the body tells right
from left. Scientific American 280 (June): 46-51.
Charlesworth, B., 2002. Evolution by Design? Nature
418: 129.
Coyne, J. A., 1996. God in the details, Nature
383: 227-228.
Darwin, C. 1872. The Origin of Species (6th
edition). London: Oxford University Press.
Dembski, W. 2002a, No Free Lunch: Why specified
complexity cannot be purchased without intelligence. Lanham, Maryland:
Rowman & Littlefield.
Dembski, W., 2002b. Detecting design in the natural
sciences, Natural History 111 (April): 76.
Depew, D. J. (1998), Intelligent design and irreducible
complexity: A rejoinder. Rhetoric and Public Affairs 1: 571-578.
Doolittle, R. F., 1993. The evolution of vertebrate
blood coagulation: A case of yin and yang, Thrombosis and Heamostasis
70: 24-28.
Doolittle, R. F., and D. F. Feng, 1987. Reconstructing
the evolution of vertebrate blood coagulation from a consideration of
the amino acid sequences of clotting proteins. Cold Spring Harbor Symposia
on Quantitative Biology 52: 869-874.
Forrest B., 2002. The newest evolution of creationism.
Natural History 111 (April): 80.
Gura, T., 2002, Evolution Critics seek role for
unseen hand in education. Nature 416: 250.
Heuck, C. J., 1998. Type III protein secretion
systems in bacterial pathogens of animals and plants, Microbiol. Mol.
Biol. Rev. 62: 379-433.
Holden, C., 1999. Kansas Dumps Darwin, Raises Alarm
Across the United States. Science 285: 1186-1187.
Huynen, M. A., T. Dandekar, and P. Bork (1999)
Variation and evolution of the citric-acid cycle: a genomic perspective.
Trends in Microbiology 7: 281-291.
McNab, R. M., 1999. The Bacterial Flagellum: Reversible
Rotary Propellor and Type III Export Apparatus. Journal of Bacteriology
181: 7149—7153.
Melendez-Hevia, E., T. G. Wadell, and M. Cascante,
1996. The puzzle of the Krebs citric acid cycle: Assembling the pieces
of chemically feasible reactions, and opportunism in the design of metabolic
pathways during evolution. J. Molecular Evolution 43: 293—303
Miller, K. R. 1999. Finding Darwin's God.
New York: Harper Collins.
Miller, K. R., 1996. A review of Darwin's Black
Box, Creation/Evolution 16: 36-40.
Orr, H. A., 2002. The Return of Intelligent Design,
The Boston Review (Summer 2002 Issue).
Padian, K., 2002. Waiting for the Watchmaker, Science
295: 2373-2374.
Pennock, R. T., 2001. Intelligent Design Creationism
and Its Critics: Philosophical, Theological, and Scientific Perspectives.
Cambridge, Massachusetts: MIT Press.
Robinson, A. J., M. Kropatkin, and P. M. Aggeler,
1969. Hagemann Factor (Factor XII) Deficiency in Marine Mammals. Science
166: 1420-1422.
Schneider, T.D. (2000), Evolution of biological
information. Nucleic Acids Research 28: 2794-2799.
Stern, C., 2002. Embryology: Fluid flow and broken
symmetry, Nature 418: 29-30.
Thewissen, J. G, M., E. M. Williams, L. J. Roe,
and S. T. Hussain, 2001. Skeletons of terrestrial cetaceans and the relationship
of whales to artiodactyls, Nature 413: 277-281.
Thewissen, J. G. M., S. T. Hussain, and M. Arif,
1994. Fossil evidence for the origin of aquatic locomotion in archaeocete
whales, Science 286: 210-212.
Thornhill, R. H., and D. W. and Ussery, 2000. A
classification of possible routes of Darwinian evolution, The Journal
of Theoretical Biology 203: 111-116.
Wells, J, 2002. Elusive Icons of Evolution, Natural
History 111 (April): 78.
Yonekura, K., S. Maki, D. G. Morgan, D. J. DeRosier,
F.Vonderviszt, K.Imada, and K. Namba, 2000. The Bacterial Flagellar Cap
as the Rotary Promoter of Flagellin Self-Assembly, Science 290:
2148-2152.
Return
to Ken Miller's Home Page


